Our supplier Technobis recently held a webinar regarding effective investigation of crystallization kinetics for process design and control of polymorphic systems. Read more about the webinar below and look at the webinar in the recording.
Webinar topic
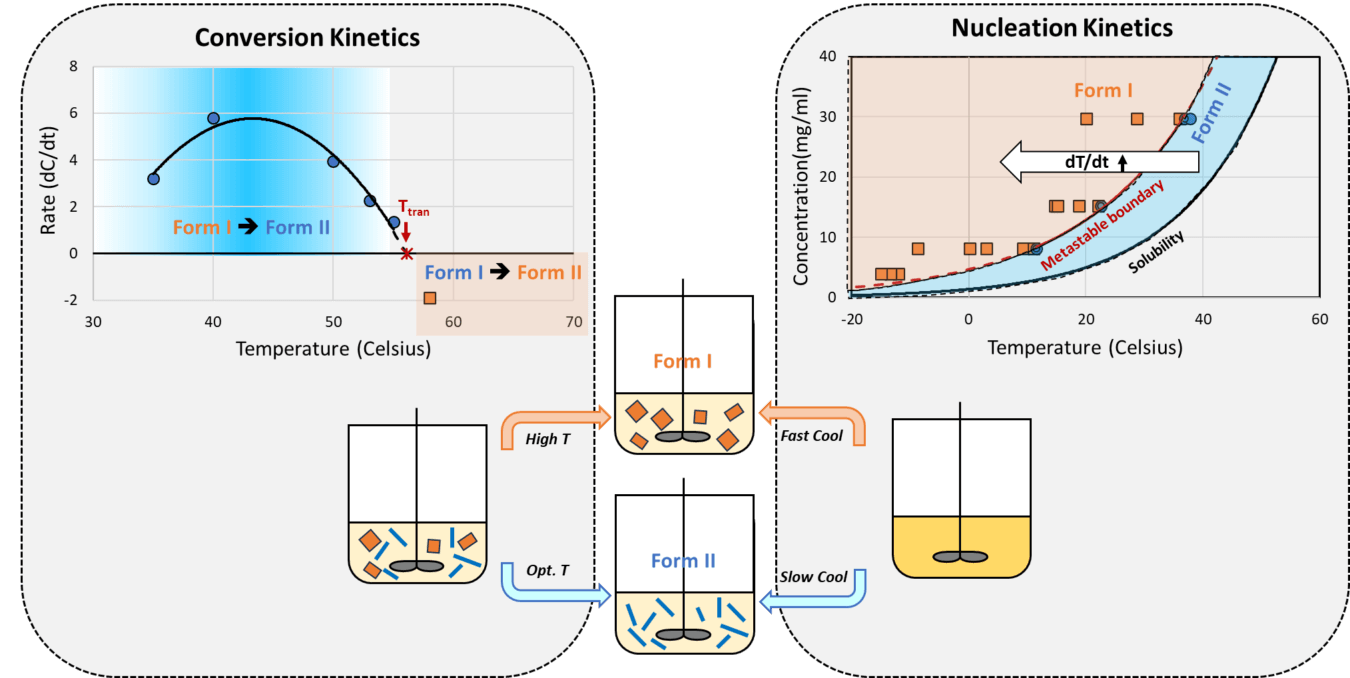
In isolation of a drug substance or its intermediate, either a more stable or a metastable form can be the target polymorph of product. It can be very challenging to ensure robust isolation of pure target form directly from a crystallization process. In this webinar, we present practical methodologies that allow collection of kinetic data to guide effective process design and control for robust isolation of either form of an enantiotropic pair.
Through a case study, we will illustrate how combined assessment of polymorphic conversion kinetics and nucleation kinetics via applications of PAT tool and automation can provide critical data that are difficult to obtain otherwise. Such data indicate proper ranges of critical process parameters and offer direct guidance for rational process design to target either the more stable or the metastable polymorph.